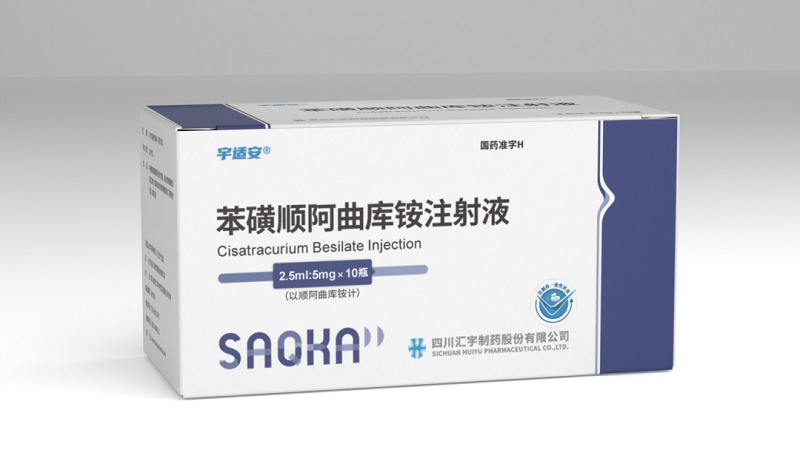
Cisatracurium Besilate 10?mg / 5?mL Vial Wholesale price comparison
$20.7
$32.91
Description Contents hide 1 Product Specifications 2 Mechanism of Action 3 Research Applications & Pharmacology 4 Safety Profile & Handling 4.1 ?? Research?Use Disclaimer Product SpecificationsAttributeDetailsProduct NameCisatracurium Besilate InjectionGeneric NameCisatracurium Besilate (Nimbex®)CAS Number96946?42?8Molecular FormulaC??H??N?O??S? (besilate salt)Molecular Weight~1243.5?g/mol en.wikipedia.org 4accessdata.fda.gov 4en.wikipedia.org 4pdr.net 2en.wikipedia.org 2pfizermedicalinformation.com 2Dosage FormAqueous solution for IV injectionStrength10?mg per 5?mL vial (2?mg/mL)Pack Size1 vial per boxApproval NumberH20183042 (China NMPA)Product Code86901445002935ManufacturerJiangsu Hengrui Pharmaceuticals Co., Ltd.Barcode(Not specified)StorageRefrigerate at 2–8?°C; protect from lightIntended UseLaboratory research onlyMechanism of ActionCisatracurium competitively binds to nicotinic cholinergic receptors at the neuromuscular junction, preventing acetylcholine binding and blocking muscle contraction. It is metabolized via Hofmann elimination—organ-independent—making it suitable for varied preclinical models.Research Applications & PharmacologyMuscle relaxation models: Suitable for tracheal intubation, ventilation, and anesthesia simulation .Pharmacokinetics: Onset ~2–4?min; effective duration ~55?min; half-life ~20–30?min; slower onset in geriatrics or renal impairment .Comparative profile: Less histamine release and hemodynamic impact than atracurium, with faster recovery compared to vecuronium.Safety Profile & HandlingAdverse events: Minimal histamine-mediated reactions (flushing, hypotension); rare bronchospasm .Hypersensitivity: Anaphylaxis reported—emergency protocols required .Lab use guidance: Administer IV with neuromuscular monitoring (e.g., train-of-four), maintain ventilatory support, have resuscitation and reversal agents available .?? Research?Use DisclaimerThis product is intended exclusively for laboratory research. It is not approved for therapeutic, diagnostic, clinical, or veterinary use. Misuse may pose safety hazards and compromise study validity.
Pain Relief Research