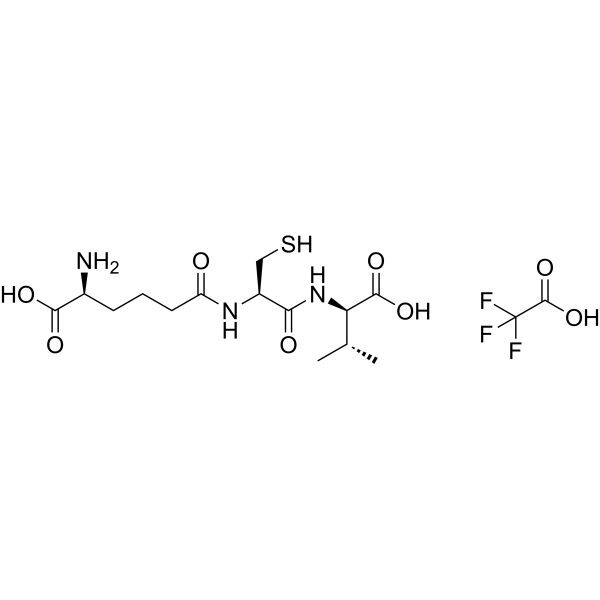
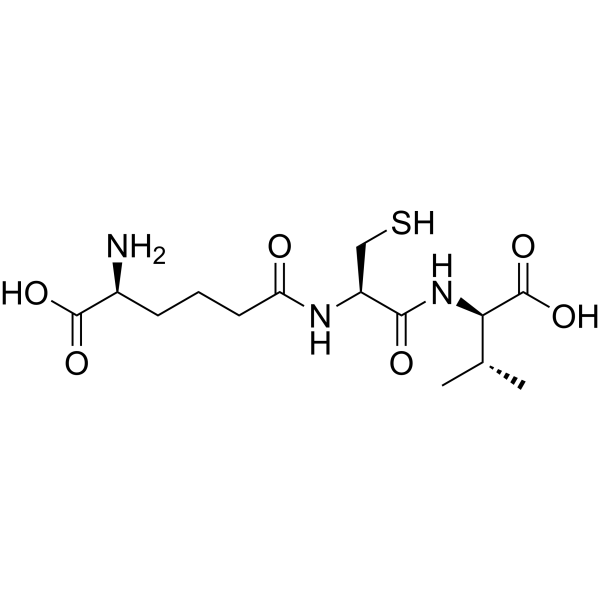
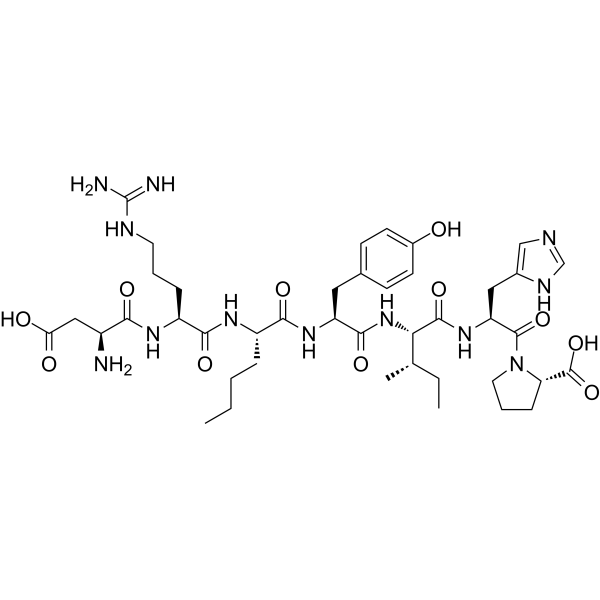
ACV Tripeptide | CAS No. 32467-88-2
$20.7
$36.64
Description Contents hide 1 Product Description 1.1 Biosynthetic Context 1.2 Significance in β-Lactam Antibiotics 1.3 Research Applications 1.4 Importance in Modern Medicine 2 Product Specifications 2.1 Notes 3 Mechanism of Action 3.1 Stepwise Role in β-Lactam Biosynthesis 3.2 Research Impact 4 Side Effects 4.1 Observed in Research 4.2 Potential Risks 4.3 Laboratory Safety Notes 5 Disclaimer 6 Keywords 7 Shipping Guarantee 8 Transaction Guarantee Product DescriptionACV Tripeptide (CAS 32467-88-2) is a non-ribosomal peptide consisting of L-α-aminoadipic acid, L-cysteine, and L-valine (ACV). It is the direct precursor to the β-lactam nucleus, which forms the structural core of penicillin and cephalosporins, two of the most clinically important classes of antibiotics.Biosynthetic ContextThe biosynthesis of ACV Tripeptide occurs in fungi such as Penicillium chrysogenum and Acremonium chrysogenum. This tripeptide is assembled by the enzyme ACV synthase, a non-ribosomal peptide synthetase (NRPS) encoded by the pchAB gene cluster (~11 kb). ACV synthase catalyzes the stepwise condensation of the three amino acids:L-α-aminoadipic acidL-cysteineL-valineThe resulting tripeptide is then cyclized to form isopenicillin N by isopenicillin N synthase (IPNS), a pivotal step in the β-lactam antibiotic pathway.Significance in β-Lactam AntibioticsPenicillins: ACV Tripeptide provides the foundation for the thiazolidine and β-lactam rings.Cephalosporins: Extended biosynthetic steps modify the ACV backbone into cephalosporin precursors.Industrial fermentation: The regulation of ACV synthase expression directly influences antibiotic yields.Research ApplicationsAntibiotic Biosynthesis Research: ACV Tripeptide is central to understanding how fungi synthesize β-lactam antibiotics.Genetic Engineering: Modifying pchAB genes allows strain optimization for enhanced antibiotic production.Metabolic Pathway Studies: ACV levels are biomarkers of fungal productivity in penicillin and cephalosporin biosynthesis.Synthetic Biology: Serves as a model for engineering novel antibiotics by manipulating non-ribosomal peptide synthases.Importance in Modern MedicineSince the discovery of penicillin, β-lactam antibiotics have remained the cornerstone of antibacterial therapy. Understanding and manipulating the biosynthesis of ACV Tripeptide has contributed to industrial advances, improving yields and enabling semi-synthetic derivatives with broader activity.Product SpecificationsItemDetailsProduct NameACV TripeptideCAS No.32467-88-2Synonymsδ-(L-α-aminoadipyl)-L-cysteinyl-D-valine, ACV precursorMolecular FormulaC14H24N4O7SMolecular Weight392.43 g/molStructureTripeptide of α-aminoadipic acid, cysteine, valineSourceBiosynthesized in Penicillium and Acremonium spp.Purity≥95% (HPLC)AppearanceWhite to off-white lyophilized powderSolubilitySoluble in water, buffers, DMSOStorage-20°C, desiccated, away from lightStabilityStable for 12 months under recommended conditionsApplicationsPenicillin and cephalosporin biosynthesis research, metabolic engineering, antibiotic pathway studiesNotesProduced in trace amounts naturally, but can be synthesized chemically for research.Analytical validation: confirmed by HPLC and MS.Critical intermediate in antibiotic fermentation industry.Mechanism of ActionACV Tripeptide itself is a biosynthetic intermediate rather than a pharmacologically active compound. Its mechanism of action relates to its role as a precursor molecule in fungal biosynthetic pathways.Stepwise Role in β-Lactam BiosynthesisNon-ribosomal synthesisACV synthase activates and links aminoadipic acid, cysteine, and valine in an ATP-dependent process.This reaction bypasses ribosomal machinery, using NRPS enzyme modules.Formation of ACV TripeptideThe enzyme arranges the peptide in a specific order, ensuring the correct stereochemistry.Cyclization to Isopenicillin NIsopenicillin N synthase (IPNS) catalyzes oxidative cyclization, generating the β-lactam and thiazolidine rings.Branching PathwaysIn penicillin biosynthesis, isopenicillin N is modified to penicillin G or V.In cephalosporin biosynthesis, additional enzymes (expandase, hydroxylase) convert it into cephalosporins.Research ImpactMetabolic Bottleneck: ACV availability is often the rate-limiting step in penicillin production.Genetic Engineering: Overexpression of pchAB enhances ACV levels and increases antibiotic yields.Synthetic Analogs: Modified tripeptides can alter antibiotic activity profiles.Side EffectsSince ACV Tripeptide is not used clinically but only in laboratory research, its safety profile is largely experimental.Observed in ResearchStable and non-toxic under standard laboratory conditions.Does not show pharmacological activity in mammalian cells.Potential RisksMay act as an irritant if inhaled or exposed to skin/eyes.Handling requires gloves and protective equipment.Long-term effects are not established as it is not intended for human administration.Laboratory Safety NotesClassified as for research use only.Avoid ingestion or injection.Dispose of according to institutional chemical waste protocols.DisclaimerThis product is intended for research purposes only. ACV Tripeptide is not approved for clinical use in humans or animals.KeywordsACV Tripeptide, CAS 32467-88-2, penicillin biosynthesis precursor, cephalosporin biosynthesis, ACV synthase, pchAB gene, β-lactam antibiotics research, non-ribosomal peptide synthesis, fungal secondary metabolism, antibiotic precursor peptide.Shipping GuaranteeWe provide secure international delivery:Customs-Cleared & Tax-Inclusive shipping available.100% Compensation for transit damage or loss.Trackable and Efficient Delivery worldwide.Transaction GuaranteeWe support multiple safe payment methods:T/T bank transferPayPalCryptocurrency (BTC, ETH, USDT, etc.)More available on requestEnsuring a safe, transparent, and convenient procurement process.
Peptide Research