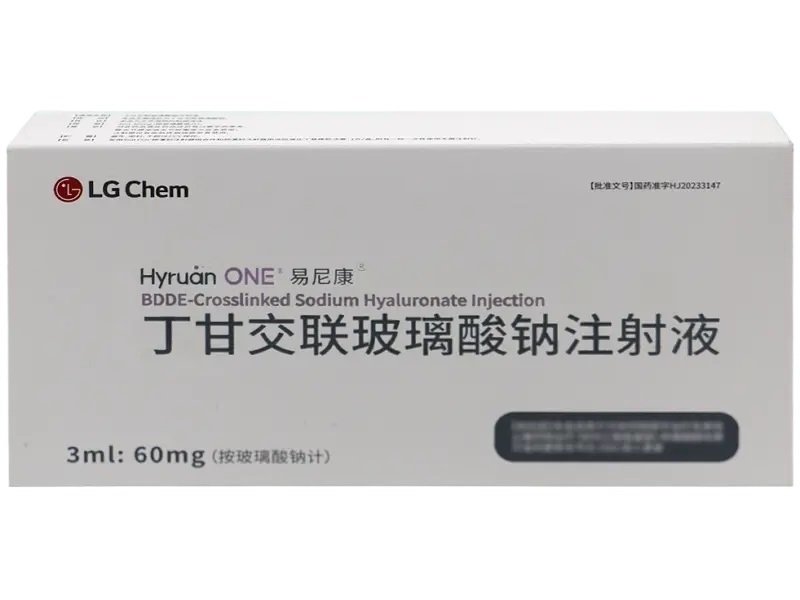
BDDE-Cross Linked Sodium Hyaluronate Injection – 60mg Prefilled Syringe
$28.76
$55.51
DescriptionBDDE-Cross Linked Sodium Hyaluronate is a chemically stabilized hyaluronic acid designed to enhance bio-stability and viscoelastic properties. This product is widely used in joint lubrication, cartilage protection, and soft tissue regeneration research.The formulation employs 1,4-butanediol diglycidyl ether (BDDE) as a crosslinker, creating a three-dimensional network that resists enzymatic degradation. It mimics synovial fluid to improve mobility and comfort in experimental osteoarthritis models.This injection comes in a prefilled syringe (3ml:60mg) with a sterile needle, sealed for safety and precision. Manufactured by LG Chem, Ltd. (South Korea) and registered under China NMPA HJ20233147, product code 86981788000164.?? Strictly for laboratory research use only. Not intended for human or animal clinical application. Contents hide 1 BDDE-Cross Linked Sodium Hyaluronate Injection Product Specifications 2 Mechanism & Research Applications 3 BDDE-Cross Linked Sodium Hyaluronate Injection Side Effects (Observed in Preclinical Studies) 4 Safety & Handling 5 Core Keywords 6 Research Use Disclaimer BDDE-Cross Linked Sodium Hyaluronate Injection Product SpecificationsParameterDetailsProduct NameBDDE-Cross Linked Sodium Hyaluronate InjectionGeneric NameSodium Hyaluronate (crosslinked with BDDE)CAS Number9067-32-7 (Hyaluronic Acid)Molecular Formula(C14H21NO11)nMolecular WeightVariable, depends on crosslinkingFormulationInjection, prefilled syringe with sterile needleStrength60mg/3mlQuantity1 syringe per boxApproval NumberHJ20233147 (China NMPA)Product Code86981788000164ManufacturerLG Chem, Ltd. (South Korea)BarcodeNot officially recordedUseLaboratory research onlyStorage ConditionsStore at 2°C–25°C; avoid direct sunlight and freezing Mechanism & Research ApplicationsBDDE-crosslinked hyaluronic acid resists enzymatic breakdown by forming ether bonds between polymer chains. This stabilization enhances viscosity, elasticity, and persistence, making it ideal for research involving:Osteoarthritis models (OA)Intra-articular lubrication studiesCartilage regeneration and protectionSynovial fluid replacement researchTissue engineering and 3D bioprinting supportBDDE-Cross Linked Sodium Hyaluronate Injection Side Effects (Observed in Preclinical Studies)Although not intended for clinical use, preclinical and in vivo models may show:Local swelling or inflammation at injection siteTemporary joint stiffness or mild painRare hypersensitivity to crosslinkers (BDDE)No systemic toxicity observed under standard dosingSafety & HandlingCategoryGuidelinesIntended UseLaboratory research only (in vitro/in vivo)Not For Use InHumans or animals in therapeutic/diagnostic contextsProtective GearGloves, lab coat, and safety goggles required during handlingStorageRefrigerated (2–8°C) or below 25°C if labeled; avoid freezingDisposalDispose according to local chemical and biological waste protocolsCore KeywordsBDDE-cross linked sodium hyaluronate, hyaluronic acid injection 60mg 3ml, LG Chem BDDE HA, BDDE hyaluronic acid research, crosslinked sodium hyaluronate lab grade, osteoarthritis joint injection study, injectable HA for research, prefilled syringe sodium hyaluronateResearch Use DisclaimerThis product is intended solely for scientific research purposes. It is not approved for human therapeutic, diagnostic, or cosmetic use. Any misuse may result in regulatory or health risk. Users must follow institutional biosafety protocols and regional regulations.
Orthopedic Research