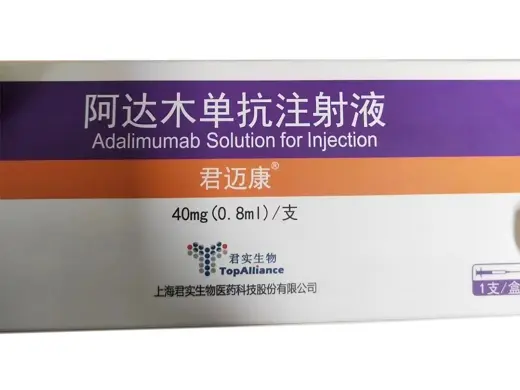
Adalimumab Injection (Sulinnuo® by Xinda Biopharma) 40?mg Prefilled Syringe
$17.41
$32.21
DescriptionAdalimumab is a fully human monoclonal antibody targeting tumor necrosis factor?? (TNF??), a key cytokine in inflammatory and autoimmune pathways. Sulinnuo® is Xinda Biopharma’s domestically produced biosimilar equivalent to Humira®, supplied in a 40?mg/0.8?ml prefilled syringe format for precise dosing.The product is manufactured by Xinda Biopharma (Suzhou) and received Chinese NMPA approval S20200020. It is commonly utilized in research projects modeling rheumatoid arthritis, Crohn’s disease, psoriasis, and other TNF-mediated inflammation processes.?? For laboratory research use only. Not intended for clinical, veterinary, or diagnostic use. Contents hide 1 Adalimumab Injection Product Specifications 2 Adalimumab Injection Mechanism & Research Applications 3 Adalimumab Injection Side Effects (Observed in Preclinical/Research Settings) 4 Safety & Handling 5 Core Keywords 6 Research Use Disclaimer Adalimumab Injection Product SpecificationsParameterDetailsProduct NameAdalimumab Injection (Sulinnuo®)Generic NameAdalimumabMab TypeFully human anti?TNF?? IgG1 monoclonal antibodyFormulationPrefilled syringe (subcutaneous injection)Strength40?mg in 0.8?mlPackagingOne syringe per boxApproval NumberS20200020 (NMPA)ManufacturerXinda Biopharma (Suzhou), ChinaIntended UseLaboratory research onlyStorage ConditionsStore refrigerated at 2–8?°C; protect from lightAdalimumab Injection Mechanism & Research ApplicationsAdalimumab selectively binds TNF??, neutralizing its pro-inflammatory effects and preventing receptor activation. Sulinnuo® is employed in research for:Autoimmune disease models (RA, Crohn’s, psoriasis, uveitis)Inflammatory signaling and cytokine cascade studiesComparative efficacy of biosimilars vs originatorsPreclinical evaluation of TNF?? blockade mechanismsAdalimumab Injection Side Effects (Observed in Preclinical/Research Settings)Based on clinical biosimilar data, research models may show:Increased susceptibility to infections (e.g. TB, fungal, bacterial)Infusion or injection site reactions (fever, chills, hypotension)Laboratory anomalies: elevated liver enzymes, cytopeniasAutoimmune phenomena akin to lupus-like syndromeRare risks: demyelinating events, lymphoma (high-dose/prolonged exposure)Recommendation: Monitor immunological markers and animal health.Safety & HandlingUse Restriction: For laboratory research onlyPPE: Use gloves, lab coat, and safety eyewearStorage: Store at 2–8?°C, avoid freezing or direct lightPreparation: Use aseptic technique during handlingDisposal: Follow institutional biohazard guidelinesCore KeywordsAdalimumab Sulinnuo injection, Xinda Biopharma adalimumab, 40?mg prefilled TNF?? antibody, S20200020 biosimilar adalimumab, lab-grade adalimumab syringe, autoimmune research biologic, wholesale adalimumab, TNF?? inhibitor for researchResearch Use DisclaimerThis product is exclusively for laboratory research. It is not approved for clinical, therapeutic, diagnostic, or veterinary use. Misuse may result in health risks and regulatory penalties. Users must follow biosafety and chemical handling protocols as per institutional guidelines.
Orthopedic Research