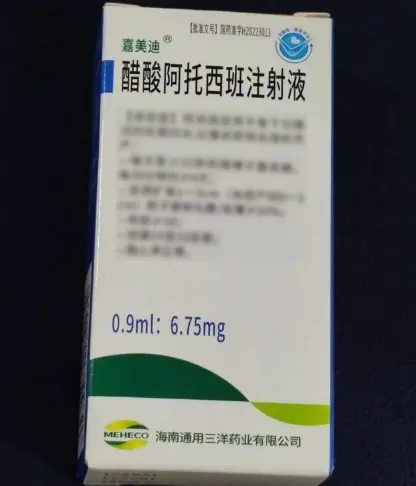
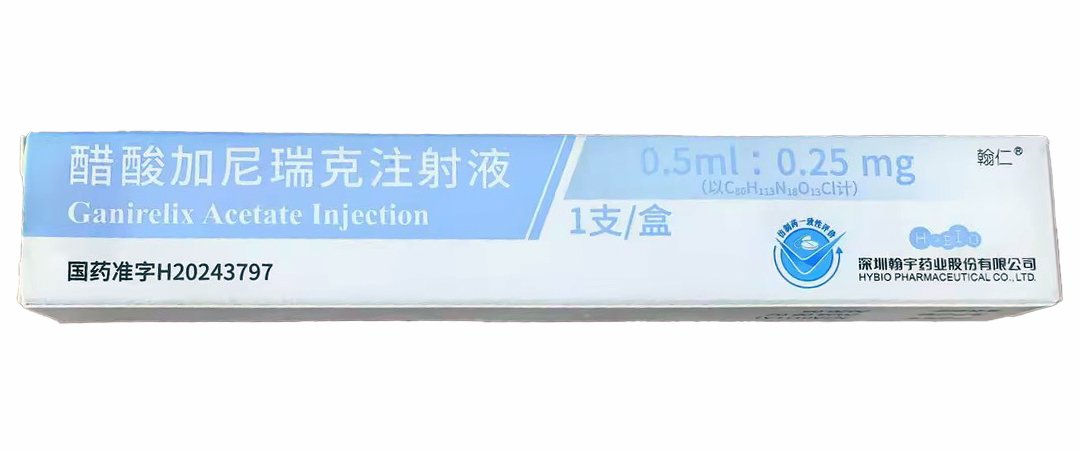
Ganirelix Acetate Injection 0.5?mL:0.25?mg
$26.87
$34.39
Description Contents hide 1 Ganirelix Acetate Injection Product Specifications 2 Ganirelix Acetate Injection Mechanism of Action 3 Research Applications & Pharmacology 4 Safety Profile & Handling 5 ?? Research Use Disclaimer Ganirelix Acetate Injection Product SpecificationsParameterDetailsProduct NameGanirelix Acetate InjectionGeneric NameGanirelix AcetateFormulationInjectable solution (prefilled syringe)Strength0.25?mg in 0.5?mLPack Size1 syringe per unitDosage FormSubcutaneous injectionApproval NumberNMPA China: H20243469Product Code86983233000033ManufacturerSuzhou Erye Pharmaceutical Co., Ltd.CAS Number124904?93?4Barcode6955872600169StorageStore at 20–25?°C (room temp); protect from lightIntended UseLaboratory/fertility research onlyGanirelix Acetate Injection Mechanism of ActionGanirelix acetate is a GnRH antagonist that competitively binds GnRH receptors on pituitary gonadotrophs, rapidly suppressing LH and FSH release to prevent premature LH surge during ovarian stimulation fda.report 2accessdata.fda.gov 2en.wikipedia.org 2.Research Applications & PharmacologyControlled ovarian hyperstimulation (COH) modelsFertility and reproductive biology assaysHormonal regulation and LH/FSH suppression studiesPharmacokinetics (250?µg dose):Tmax ~1.1?h, half-life ~16.2?h, 91% bioavailabilityEliminated ~75% in feces, ~22% in urineSafety Profile & HandlingCommon Adverse Reactions (in clinical trials):Injection site pain, redness, swellingAbdominal/gastrointestinal pain, nausea, vaginal bleeding, headachesRare ovarian hyperstimulation syndrome (OHSS)Handling & Storage:Use lab PPE: gloves, mask, coatFor subcutaneous injection onlyStore at 20–25?°C, protect from lightNot for human/veterinary use — for lab protocols only?? Research Use DisclaimerThis product is intended solely for laboratory research use. Not authorized for human, veterinary, clinical, or diagnostic purposes. Misuse may violate regulations and pose health risks.
Gynecological Research